
In ancient Babylon, Egypt, Greece, and Rome, soap was being made by boiling animal fats and wood ashes in water. This practice was probably imported to Europe around the 6th century BC by the Phoenicians, merchants who used soap to wash the fabrics they traded for other goods. Despite this, the use of soap for personal hygiene is not mentioned until the second century in the writings of Galen, the most accomplished medical researcher during Antiquity. Galen also noted that soap had a number of benefits for the skin. Contrary to popular belief, soap production and use were widespread during the Middle Ages, particularly among higher social classes. Spain produced the most soap during the ninth century, later to be followed by Marseilles, Genoa, Venice, and Savona, places rich in oil and caustic soda. But by the late Middle Ages, the use of soap declined when people turned to perfumes to cover up their smells. People only started using soap again regularly for personal hygiene in the nineteenth century.
1) MATERIAL
- 1 glass bowl, non-aluminum pot, or earthenware container
- 1 wooden spoon
- 1 wooden case
- ¼ liter of oil (approximately 1 glass)
- ¼ liter of water
- 3 teaspoons of caustic soda (also called sodium hydroxide or lye)
- Goggles
- Rubber gloves

CAUTION: caustic soda is highly corrosive! You must take security measures and avoid contact with skin or clothes. If this should happen, wash with abundant water and soap.
2) LET’S GET CRACKING!
- Before you start, put on your goggles and rubber gloves.
- Place caustic soda in the container, and pour water slowly. Be extremely careful not to touch the soda with your hand as it may burn your skin. While preparing this solution, you will see that it gives off heat: this type of reaction is called exothermic.
- Pour in the oil slowly, stirring gently with the wooden spoon for quite some time. You will be done once a whitish paste begins to thicken (be patient!).
- If you continue to stir but the paste does not thicken, don’t get rid of it. Pour it in a pot and heat it up on the stove. Stir again and the soap will begin to appear.
- To make the soap whiter, you can add a bluing agent, like blue pigment from indigo. To make scented soap, add a fragrance or essential oil (lemon), or even a sprinkling of aromatic herbs.
- With the help of a spatula allow the soap to drain on a paper towel. Then, use a spatula to transfer the paste to a wooden mold and let it sit 24 to 48 hours. Then, unmold the soap and cut it into bars using a sharp knife. It is now ready to be used!
3) WHAT HAPPENED?
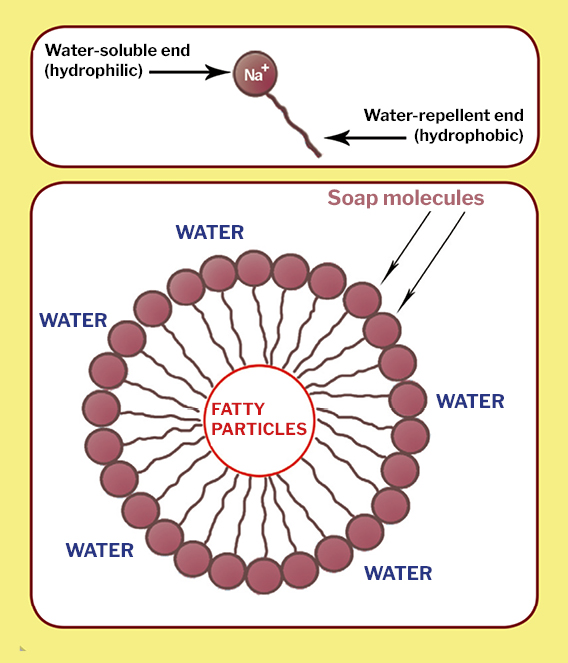
Oils are formed of long molecules called fatty acids. These molecules, called hydrophobic (“scared” of water”), don’t mix with water. When lye is mixed with oil and water, the soda releases sodium cations (positively-charged sodium atoms or Na+), and these attach to the end of fatty acids. As a result, an asymmetrical molecule forms: one end of the chain is water soluble (hydrophilic) and the other end is not (hydrophobic). These are the ever so peculiar soap molecules.
4) HOW DOES SOAP WORK?
When soap comes into contact with water, the soap molecules arrange into minute clusters called micelles. The hydrophilic part of the molecule points outwards, forming the outer surface of the micelle, whereas the hydrophobic part groups on the inside. The fats (or fatty particles) in the stains are attracted to the inner hydrophobic part and separate from the surface of the fabric or skin. This creates an emulsion (fatty molecules held in suspension in the water). The emulsion washes out with water.











Leave a Reply